

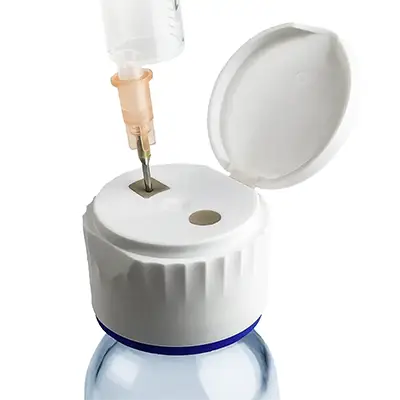
Septum-Lid Cap
This innovative dual-component closure combines a convenient flip-top lid with an integrated, injection-molded septum that reliably self-seals after each needle puncture.


CHAMELEON® Cap
The CHAMELEON® cap from KISICO is a transparent desiccant closure with a built-in moisture indicator. The color change from amber to light yellow immediately indicates the moisture level—no need to open the package, no uncertainty. For product safety and less waste throughout the supply chain.



Evaporation Stopper
Our evaporation stoppers prevent unwanted evaporation, reduce the risk of contamination, and the entry of foreign particles, thereby ensuring high sealing integrity.



LyLift System
Combines the LyStopper and the LyCap into one – clicked and fused securely into place during lyophilization. As a result, both LyCap and LyStopper can be lifted out as one unit.



Wide-neck Caps
Screw caps for wide-necked glasses and jars – premium design and high-quality product produced in class 8 cleanroom



Two-component caps without port
Closures with injected temperature-resistant sealing inserts and with injected chemical-resistant sealing cones.



Two-component caps with port
Caps featuring injected seals as well as pierceable ports. Self-resealing properties even after several uses.



Recyclable caps
Caps made from monomaterials that can be recycled well in the circular economy.



Components
Technical components produced to customer specifications or specially developed injection-molded parts.



Packaging
Snap-on caps in different designs, e.g., for containers with spray pumps or dispensers. Available in a range of colors.



Special packaging
We are happy to implement customized packaging requirements such as multiple bags, indicators, and custom labels.



Liners, gaskets, rings
Liners and gaskets made from foamed PE, rubber, TPE and many other materials. Available in in a variety of diameters and thickness.



Caps made from recyclates
Caps made from renewable sources such as recycled or biodegradable materials, among other innovative and sustainable substances.


Caps made from renewables
Caps produced from renewable and sustainable sources from base materials such as bagasse, wood, starch, or milk.



Sterilization
We work with certified sterilization partners to offer gamma-, hot-, and EtO sterilization for our caps.



Venting caps
Caps with membranes that allow pressure equalization, effectively preventing the container from bulging or buckling.



Customized solutions
If you can´t find what you are looking for in our catalog, we are happy to design a suitable closure for your application.


DIN Caps
Closures with sealing cone, sealing lip, or sealing liners for standard screw threads such as DIN GL, GCMI et al.



Caps with center opening
Closures with a center opening that can be used, for example, as coupling connections and pipette closures.



Double-walled caps
Double-walled closures for high-value packaging design. Spouts or sealing discs may optionally be included.



Hinged caps
Easy-to-open, durable closures with hinged lids. Available in a variety of colors and materials to suit a range of applications.


Snap-on caps
Snap-on caps in different designs, e.g., for containers with spray pumps or dispensers. Available in a range of colors.

Bottles
Bottles produced as per our clients´ individual requirements to the highest standards under a monitored production environment.



Pierceable septa
Pierceable septa with customizable features such as design, hardness grade, temperature-resistance, and color.



Measuring spoons
Single or double spoons with one or two measuring scales for accurate dosing of a wide range of substances.



Threaded measuring cups
Screwcaps and snap-on caps with a measuring scale for precise and clean dosing of a wide variety of substances.


Measuring cups
Measuring cups in a range of volumes. Can be made from different materials. Scaling can be embossed or printed. Can be delivered with the CE mark.



Caps with brush
Screwcaps with assembled brushes. The materials and brushes can be individually customized to suit.



Dosing caps
Screwcaps with droppers, dispensers as well as pourers for the precise dosing of all kinds of fluids.



Child-resistant caps
Caps produced in cleanroom conditions from materials that meet the highest quality and regulatory standards.



Caps with desiccant
Caps with integrated desiccant chamber. Volume and type of desiccant can be adjusted to suit.



Tamper-evident caps
Caps with tamper-evident rings for first-class product protection. Can be combined with child-resistant and desiccant functions.

Cleanroom
Caps produced in cleanroom conditions from materials that meet the highest quality and regulatory standards.



Vial Closures
Screw caps for vials available in a variety of colors. Suitable for most of the common thread sizes.



Bottle Caps
Screw caps for bottles that can be equipped with gaskets for a secure seal in the packaging of chemicals, media, or food.



Caps for laboratories
Temperature- and chemical-resistant caps as well as screw-thread couplings for a variety of laboratory applications.



Biodegradable caps
Caps produced from materials that are certified according to international standards to be biodegradable.



Caps with integrated NFC tag
Our revolutionary NFCCap enables contactless communication with any NFC-enabled smartphone. For phones without an NFC reader there are corresponding add-on devices.
This technology can be built into most closures from KISICO’s standard range. This avoids the costs for new injection molds or mold modification. The closures retain their design because the chip is not visible from the outside. One advantage for users in the food, chemical and pharmaceutical industries, is that the chip does not come into contact with the filling material.
A wide range of information and commands can be programmed onto the chip. Appropriate applications can be used to verify that the product is not a counterfeit. This technology can also be used to communicate with a tech-savvy target group. Links to websites, product data sheets or specially developed apps can be opened by communication between the closure and smartphone.



Multicomponent caps
Caps produced using multicomponent injection-molding techniques, e.g. caps with injected port covers.



TPE Stopper
Latex-free, additive-free, and cleanroom-made – the smart alternative to rubber stoppers for lyophilization and sensitive applications.



Caps with dosing chamber
Demand for dosing caps is on the upswing and KISICO´s ReservoirCap is the optimal solution for those needs. These caps allow substances to be mixed immediately before use.
The ReservoirCap is supplied with a ready-to-fill container. The ingredient is simply poured into the reservoir at the top of the cap. After this simple filling process has been completed, the container is closed with a lid. The ingredient is now completely enclosed in the cavity and protected from all external influences. No other sealing equipment is required. The lid is then screwed onto a standard bottle with a PP28 mouth. This bottle can be filled with a liquid. If required, the reservoir can also be filled after the ReservoirCap has been screwed onto a bottle filled with liquid. Turning the smaller cap opens the container and the substance from the reservoir falls into the bottle.
The closures can be produced in cleanroom facilities and from pharmaceutical or food-grade materials.




Caps with holograms
KISICO´s HologramCap is a trendsetting innovation. Get our existing and popular closures with an additional counterfeit security feature.
Unique breakthroughs in nanotechnology enable KISICO to offer customers unrivalled protection against counterfeiting. The design features a hologram that is fully integrated into the production process of the cap, thus requiring no additives, inks, or labels. The style of the hologram can be customized according to customer needs.
In combination with the tamper-evident ring, the HologramCap offers the most advanced protection to consumers and brand owners on the market. This solution is not only interesting for the pharmaceutical industry and others who package high-grade products. The attractive and unique appearance can also be used to differentiate between products and emphasize their high quality.


Find your product
Tags
KISICO
Kirchner, Simon & Co. GmbH
Rieslingstrasse 41
65375 Oestrich-Winkel
Germany
Phone: +49 67 23 99 65-0
E-Mail: info@kisico.de
As cap & closure experts, we have been providing optimal safety for your products and to your consumers for over 75 years. Our expertise and competence offer you a competitive advantage through customized and innovative solutions.




